Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. 1 is compulsory.
Q.1 Choose the correct option/answer the following (Any seven question
only):
According to Planck's quantum theory, energy is emitted or absorbed
In photoelectric effect, the number of photoelectrons emitted is proportional to
The mode of excitation of molecule in IR spectroscopy is
Beer-Lambert's law relates absorbance (A) with
For an ideal gas, the compressibility factor (Z) is
As one moves along a given row in the periodic table ionization energy
Waterline corrosion in steel tank is an example of
Which of the following dissolved salts in water will cause the most hardness in water sample?
Optically active stereoisomers that are not the mirror images are called
In a Diels-Alder reaction, a conjugated diene reacts with
Q.2 Solve both questions :
(i) State Heisenberg's uncertainty principle. What is uncertainty in the position of a particle,
if the uncertainty in momentum is $ 10^{-11} kg m/s $?
(ii) Explain why classical Newtonian and Maxwell's wave mechanics theory failed to describe the
behavior of microscopic (atomic and subatomic) particles.
(i) Explain the dual nature of matter and radiation. Describe de Broglie's hypothesis and briefly
discuss its experimental verification.
(ii) Explain the principles of combination of atomic orbitals to form molecular orbitals.
Q.3 Solve both questions :
Draw the molecular orbital energy level diagrams for (i) $ N_2 $ and (ii) $ O_{2} $ molecules. Write their molecular orbital electronic configurations, calculate their bond orders, and explain their magnetic properties.
(i) What are the salient features of crystal field theory? Also explain the crystal field
splitting in octahedral complexes with energy level diagram.
(ii) Differentiate between the coordination number and oxidation number of a metal ion. Further,
calculate the coordination number and oxidation number of Fe in $ K_{4}[Fe(CN)_{6}] $ and Ni
in $ Ni(CO)_4 $.
Q.4 Solve both questions :
(i) State the principle of vibrational spectroscopy and derive the selection rule for vibrational
transitions in diatomic molecules.
(ii) Explain the mechanism of fluorescence and give its important applications.
Write informative notes on any two of the following:
Q.5 Solve both questions :
(i) Derive the Van der Waals' equation of state for n moles of gas. Explain the significance of
Van der Waals' constants.
(ii) Describe the deviations of real gases from ideal behaviour and discuss their causes.
(i) Explain ionic, dipolar, and van der Waals interactions with suitable examples.
(ii) Differentiate among ionization energy, electron affinity, and electronegativity,
highlighting their definitions and periodic trends.
Q.6 Solve both questions :
(i) What is electronic configuration? Also give the electronic configuration of Cu, $ Al^{3+}
$ and $ Cl^- $.
(ii) Explain the variation of s, p, d, and f orbital energies across the periodic table.
What do you understand by hybridization? Giving suitable examples explain $ sp^{3} $, $ sp^{3}d $ and $ sp^3d^2 $ hybridization.
Q.7 Solve both questions :
(i) Predict at what temperature would the following reaction be spontaneous, given that $
\Delta H=9.080~kJ/mol $ and $ \Delta S=0.0357~kJ/(K \cdot mol) $.
$ 4NH_{3} + 5O_{2} \rightarrow 4NO + 6H_{2}O $
(ii) What is the Ellingham diagram and discuss its importance in metallurgical processes.
(i) Derive Nernst equation and write its applications.
(ii) A sample of water on analysis has been found to contain the following in ppm: $
Ca(HCO_{3})_{2}=4.86 $; $ Mg(HCO_{3})_{2}=5.84 $; $ CaSO_{4}=6.80 $; $ MgSO_{4}=8.40
$. Calculate the temporary and permanent hardness of the water.
Q.8 Solve both questions :
(i) What is principle of EDTA titration? How is the permanent hardness of water is determined
using EDTA method?
(ii) What are the ion-exchange resins? How will you purify water by using the resins? What are
the advantages of this method over other methods.
(i) What are the stereoisomers? How are they classified.
(ii) Explain the term chirality. Discuss the optical isomerism of chiral organic compounds using
the example of tartaric acid.
Q.9 Solve both questions :
(i) Draw and describe the conformations of cyclohexane. Discuss the relative stability of
conformers.
(ii) What are the different types of reactive intermediates involved in organic reactions?
(i) Discuss elimination and substitution reactions with examples.
(ii) Give a brief note on the synthesis of aspirin.
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. 1 is compulsory.
Q.1 Choose the correct option/answer the following (Any seven
only):
According to Heisenberg's Uncertainty Principle, which of the following pairs cannot be simultaneously determined with high precision?
According to Crystal Field Theory, the splitting of d-orbitals in an octahedral field leads to:
Which of the following complexes will show geometrical isomerism?
In IR spectroscopy, a vibration is IR active only if:
Which of the following is not a van der Waals force?
The spontaneity of a chemical process is indicated by:
Temporary hardness of water is mainly due to:
Which of the following elements has the highest electronegativity?
Enantiomers differ in:
Diels-Alder reaction involves
Q.2 Solve both questions :
State and explain Heisenberg's Uncertainty Principle. Discuss its significance in determining position and momentum of an electron.
Explain the photoelectric effect and de Broglie hypothesis.
Q.3 Solve both questions :
Explain the formation of molecular orbitals using the linear combination of atomic orbitals (LCAO) approach. Differentiate between bonding and antibonding orbitals.
Discuss the crystal field splitting of d-orbital in Tetrahedral complexes.
Q.4 Solve both questions :
Explain the principles of vibrational and rotational spectroscopy. What are the selection rules for rotational transitions in diatomic molecules?
Derive van der Waal's equation for real gases.
Q.5 Solve both questions :
Discuss the deviation of real gases from ideal behavior. Under what conditions is the deviation more significant?
Discuss the basic principle of Nuclear Magnetic Resonance (NMR) spectroscopy. How is it useful for structural elucidation of organic compounds?
Q.6 Solve both questions :
Explain the Nernst equation and its applications in calculating EMF of electrochemical cells.
What do you mean by Hardness of water. Discuss the EDTA method for estimation of hardness of water.
Q.7 Solve both questions :
Explain the HSAB (Hard and Soft Acids and Bases) principle with suitable examples.
Define ionization energy & electron affinity, How do these vary in the periodic table as the atomic number increases.
Q.8 Solve both questions :
Discuss the relationship between hybridization and molecular geometry. Provide examples of common geometries like tetrahedral, linear, and trigonal planar.
Define stereoisomerism. Differentiate between structural isomers, enantiomers, and diastereomers with examples.
Q.9 Solve both questions :
Describe the mechanism of epoxide ring opening reactions under acidic and basic conditions.
Outline the synthesis of aspirin from salicylic acid. Include the reagents, reaction mechanism, and significance of the reaction.
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. 1 is compulsory.
Q.1 Choose the correct option/answer the following (Any seven
only):
According to Heisenberg's Uncertainty Principle, which of the following pairs cannot be simultaneously determined with high precision?
According to Crystal Field Theory, the splitting of d-orbitals in an octahedral field leads to:
Which of the following complexes will show geometrical isomerism?
In IR spectroscopy, a vibration is IR active only if:
Which of the following is not a van der Waals force?
The spontaneity of a chemical process is indicated by:
Temporary hardness of water is mainly due to:
Which of the following elements has the highest electronegativity?
Enantiomers differ in:
Diels-Alder reaction involves
Q.2 Solve both questions :
State and explain Heisenberg's Uncertainty Principle. Discuss its significance in determining position and momentum of an electron.
Explain the photoelectric effect and de Broglie hypothesis.
Q.3 Solve both questions :
Explain the formation of molecular orbitals using the linear combination of atomic orbitals (LCAO) approach. Differentiate between bonding and antibonding orbitals.
Discuss the crystal field splitting of d-orbital in Tetrahedral complexes.
Q.4 Solve both questions :
Explain the principles of vibrational and rotational spectroscopy. What are the selection rules for rotational transitions in diatomic molecules?
Derive van der Waal's equation for real gases.
Q.5 Solve both questions :
Discuss the deviation of real gases from ideal behavior. Under what conditions is the deviation more significant?
Discuss the basic principle of Nuclear Magnetic Resonance (NMR) spectroscopy. How is it useful for structural elucidation of organic compounds?
Q.6 Solve both questions :
Explain the Nernst equation and its applications in calculating EMF of electrochemical cells.
What do you mean by Hardness of water. Discuss the EDTA method for estimation of hardness of water.
Q.7 Solve both questions :
Explain the HSAB (Hard and Soft Acids and Bases) principle with suitable examples.
Define ionization energy & electron affinity, How do these vary in the periodic table as the atomic number increases.
Q.8 Solve both questions :
Discuss the relationship between hybridization and molecular geometry. Provide examples of common geometries like tetrahedral, linear, and trigonal planar.
Define stereoisomerism. Differentiate between structural isomers, enantiomers, and diastereomers with examples.
Q.9 Solve both questions :
Describe the mechanism of epoxide ring opening reactions under acidic and basic conditions.
Outline the synthesis of aspirin from salicylic acid. Include the reagents, reaction mechanism, and significance of the reaction.
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. 1 is compulsory.
Q.1 Choose the correct option/answer the following (Any seven
only):
According to Heisenberg's Uncertainty Principle, which of the following pairs cannot be simultaneously determined with high precision?
According to Crystal Field Theory, the splitting of d-orbitals in an octahedral field leads to:
Which of the following complexes will show geometrical isomerism?
In IR spectroscopy, a vibration is IR active only if:
Which of the following is not a van der Waals force?
The spontaneity of a chemical process is indicated by:
Temporary hardness of water is mainly due to:
Which of the following elements has the highest electronegativity?
Enantiomers differ in:
Diels-Alder reaction involves
Q.2 Solve both questions :
State and explain Heisenberg's Uncertainty Principle. Discuss its significance in determining position and momentum of an electron.
Explain the photoelectric effect and de Broglie hypothesis.
Q.3 Solve both questions :
Explain the formation of molecular orbitals using the linear combination of atomic orbitals (LCAO) approach. Differentiate between bonding and antibonding orbitals.
Discuss the crystal field splitting of d-orbital in Tetrahedral complexes.
Q.4 Solve both questions :
Explain the principles of vibrational and rotational spectroscopy. What are the selection rules for rotational transitions in diatomic molecules?
Derive van der Waal's equation for real gases.
Q.5 Solve both questions :
Discuss the deviation of real gases from ideal behavior. Under what conditions is the deviation more significant?
Discuss the basic principle of Nuclear Magnetic Resonance (NMR) spectroscopy. How is it useful for structural elucidation of organic compounds?
Q.6 Solve both questions :
Explain the Nernst equation and its applications in calculating EMF of electrochemical cells.
What do you mean by Hardness of water. Discuss the EDTA method for estimation of hardness of water.
Q.7 Solve both questions :
Explain the HSAB (Hard and Soft Acids and Bases) principle with suitable examples.
Define ionization energy & electron affinity, How do these vary in the periodic table as the atomic number increases.
Q.8 Solve both questions :
Discuss the relationship between hybridization and molecular geometry. Provide examples of common geometries like tetrahedral, linear, and trigonal planar.
Define stereoisomerism. Differentiate between structural isomers, enantiomers, and diastereomers with examples.
Q.9 Solve both questions :
Describe the mechanism of epoxide ring opening reactions under acidic and basic conditions.
Outline the synthesis of aspirin from salicylic acid. Include the reagents, reaction mechanism, and significance of the reaction.
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. 1 is compulsory.
Q.1 Choose the correct option/answer the following (Any seven
only):
According to Heisenberg's Uncertainty Principle, which of the following pairs cannot be simultaneously determined with high precision?
According to Crystal Field Theory, the splitting of d-orbitals in an octahedral field leads to:
Which of the following complexes will show geometrical isomerism?
In IR spectroscopy, a vibration is IR active only if:
Which of the following is not a van der Waals force?
The spontaneity of a chemical process is indicated by:
Temporary hardness of water is mainly due to:
Which of the following elements has the highest electronegativity?
Enantiomers differ in:
Diels-Alder reaction involves
Q.2 Solve both questions :
State and explain Heisenberg's Uncertainty Principle. Discuss its significance in determining position and momentum of an electron.
Explain the photoelectric effect and de Broglie hypothesis.
Q.3 Solve both questions :
Explain the formation of molecular orbitals using the linear combination of atomic orbitals (LCAO) approach. Differentiate between bonding and antibonding orbitals.
Discuss the crystal field splitting of d-orbital in Tetrahedral complexes.
Q.4 Solve both questions :
Explain the principles of vibrational and rotational spectroscopy. What are the selection rules for rotational transitions in diatomic molecules?
Derive van der Waal's equation for real gases.
Q.5 Solve both questions :
Discuss the deviation of real gases from ideal behavior. Under what conditions is the deviation more significant?
Discuss the basic principle of Nuclear Magnetic Resonance (NMR) spectroscopy. How is it useful for structural elucidation of organic compounds?
Q.6 Solve both questions :
Explain the Nernst equation and its applications in calculating EMF of electrochemical cells.
What do you mean by Hardness of water. Discuss the EDTA method for estimation of hardness of water.
Q.7 Solve both questions :
Explain the HSAB (Hard and Soft Acids and Bases) principle with suitable examples.
Define ionization energy & electron affinity, How do these vary in the periodic table as the atomic number increases.
Q.8 Solve both questions :
Discuss the relationship between hybridization and molecular geometry. Provide examples of common geometries like tetrahedral, linear, and trigonal planar.
Define stereoisomerism. Differentiate between structural isomers, enantiomers, and diastereomers with examples.
Q.9 Solve both questions :
Describe the mechanism of epoxide ring opening reactions under acidic and basic conditions.
Outline the synthesis of aspirin from salicylic acid. Include the reagents, reaction mechanism, and significance of the reaction.
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. 1 is compulsory.
Q.1 Choose the correct option/answer the following (Any seven
only):
According to Heisenberg's Uncertainty Principle, which of the following pairs cannot be simultaneously determined with high precision?
According to Crystal Field Theory, the splitting of d-orbitals in an octahedral field leads to:
Which of the following complexes will show geometrical isomerism?
In IR spectroscopy, a vibration is IR active only if:
Which of the following is not a van der Waals force?
The spontaneity of a chemical process is indicated by:
Temporary hardness of water is mainly due to:
Which of the following elements has the highest electronegativity?
Enantiomers differ in:
Diels-Alder reaction involves
Q.2 Solve both questions :
State and explain Heisenberg's Uncertainty Principle. Discuss its significance in determining position and momentum of an electron.
Explain the photoelectric effect and de Broglie hypothesis.
Q.3 Solve both questions :
Explain the formation of molecular orbitals using the linear combination of atomic orbitals (LCAO) approach. Differentiate between bonding and antibonding orbitals.
Discuss the crystal field splitting of d-orbital in Tetrahedral complexes.
Q.4 Solve both questions :
Explain the principles of vibrational and rotational spectroscopy. What are the selection rules for rotational transitions in diatomic molecules?
Derive van der Waal's equation for real gases.
Q.5 Solve both questions :
Discuss the deviation of real gases from ideal behavior. Under what conditions is the deviation more significant?
Discuss the basic principle of Nuclear Magnetic Resonance (NMR) spectroscopy. How is it useful for structural elucidation of organic compounds?
Q.6 Solve both questions :
Explain the Nernst equation and its applications in calculating EMF of electrochemical cells.
What do you mean by Hardness of water. Discuss the EDTA method for estimation of hardness of water.
Q.7 Solve both questions :
Explain the HSAB (Hard and Soft Acids and Bases) principle with suitable examples.
Define ionization energy & electron affinity, How do these vary in the periodic table as the atomic number increases.
Q.8 Solve both questions :
Discuss the relationship between hybridization and molecular geometry. Provide examples of common geometries like tetrahedral, linear, and trigonal planar.
Define stereoisomerism. Differentiate between structural isomers, enantiomers, and diastereomers with examples.
Q.9 Solve both questions :
Describe the mechanism of epoxide ring opening reactions under acidic and basic conditions.
Outline the synthesis of aspirin from salicylic acid. Include the reagents, reaction mechanism, and significance of the reaction.
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. I is compulsory.
Q.1 Choose the correct answer the following (Any seven question
only):
How many electrons can exist with the principal quantum number's value as 4?
A ball of mass 0.5kg is moving with velocity 6.626 m/s. What's the wavelength of that ball?
As per Heisenberg's principle of uncertainty, the relation between relative momentum and relative position is ______.
Arrange the following intermediate into decreasing order of stability.
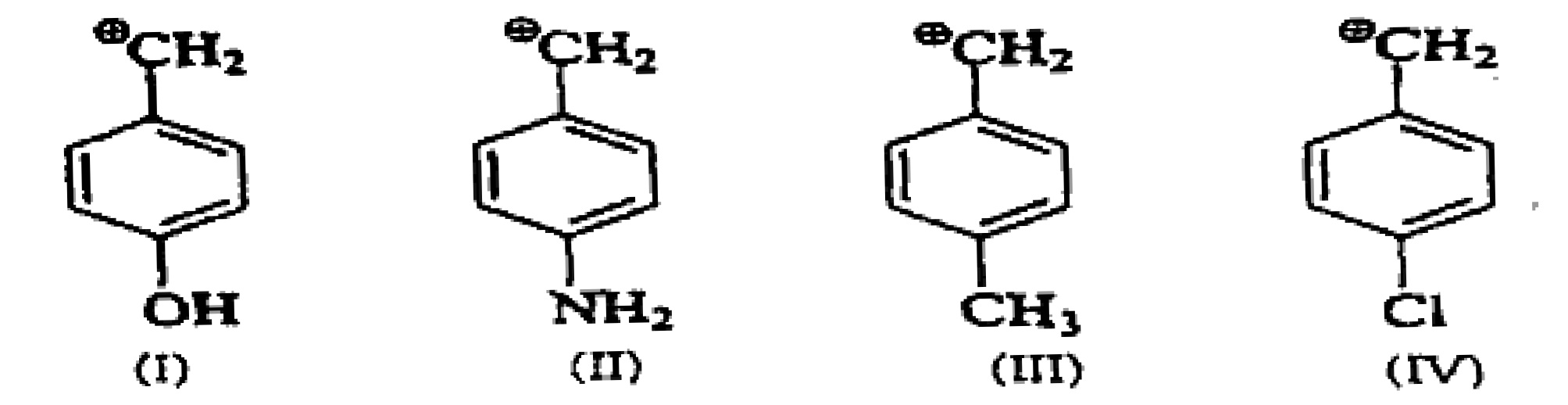
The distance between the centers of the peaks of doublet is called as?
How many peaks are expected in low-resolution NMR spectrum of vinyl chloride and ethyl cyclopropane?
Which of the following phase changes are endothermic?
The Gibbs free energy is positive when a change in enthalpy and change in entropy are positive at ______.
The type of hardness that occurs due to the presence of bicarbonate of calcium and magnesium ions hardness is ______.
Which of the following is an alkane which can exhibit optical activity?
Q.2 Solve both questions :
Write short notes on:
What are Quantum numbers and what do they signify? How are these numbers related with each other?
Q.3 Solve both questions :
Give reason for the following:-
Find out the bond orders of CO, $ \text{N}_2 $, $ \text{O}_2^\text{2-} $, $ \text{O}_2^\text{2+} $
Which of the following two molecules has a higher bond length (i) $ \text{O}_2 $: (ii) $ \text{O}_2^- $? Explain using Molecular orbital Theory.
Q.4 Solve both questions :
Describe and compare the splitting of d- orbitals under the influence of octahedral and tetrahedral ligand fields.
Explain Crystal Field Stabilization Energy (CFSE) and pairing Energy (PE). How do the CFSE values related to the corresponding colours of d-block metal complexes?
Q.5 Solve all parts :
Why is methanol a good solvent for U.V. Visible but not for IR spectroscopy?
Calculate the number of vibrational degrees of freedom in the following compounds
What is the importance of fingerprint region in IR spectra?
Q.6 Solve both questions :
Discuss spin-spin relaxation in NMR spectroscopy and draw and explain the splitting pattern observed in the 1H NMR of $ \text{CHCl}_2\text{CH}_2\text{Cl} $.
The frequency of oscillation of HF is $ 4.12 \times 10^{-14} $ Hz. Calculate the vibrational energy and its force constant. $ M_{H} = 1.0078u $, $ M_{F} = 18.9984u $.
Q.7 Solve both questions :
What are ion exchange Resins? Discuss their application in water softening. How are spent resins regenerated?
9.67ml of EDTA solution consumes exactly 10.2 mg of $ \text{CaCO}_3 $ equivalents. 100 ml of a given Hard water sample consumes 17.5 ml of above EDTA solution. 250 ml of the above hard water sample was boiled completely and boiled off sample consumed 35 ml of above EDTA solution. Calculate the temporary and permanent hardness of the above hardware sample in $ ^{\circ}C $, $ ^{\circ}F $ and as $ \text{CaCO}_3 $ equivalents.
Q.8 Solve all parts :
Differentiate between terms Configuration and Conformation with suitable examples.
Discuss stereochemistry of tartaric acid.
Explain racemisation of optical isomers with suitable examples.
Q.9 Solve both questions :
What do you mean by cyclization and ring opening reactions give two examples of each type of these reactions.
What are carbonium ions? Show hybridisation in the structure of carbonium ion and discuss the stability of primary, secondary and tertiary carbonium ions.
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. I is compulsory.
Q.1 Choose the correct answer the following (Any seven question
only):
How many electrons can exist with the principal quantum number's value as 4?
A ball of mass 0.5kg is moving with velocity 6.626 m/s. What's the wavelength of that ball?
As per Heisenberg's principle of uncertainty, the relation between relative momentum and relative position is ______.
Arrange the following intermediate into decreasing order of stability.
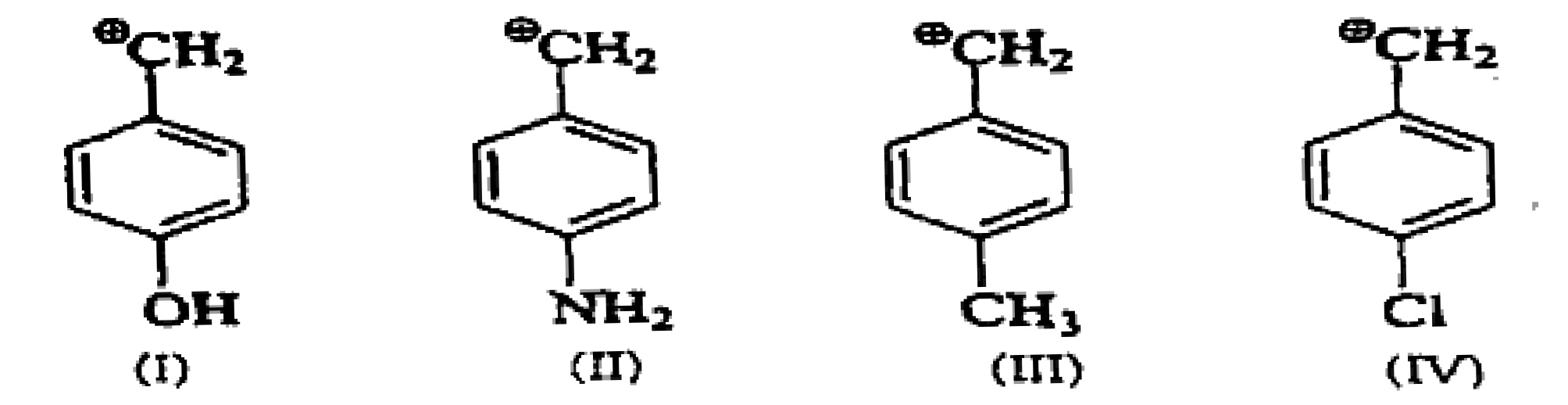
The distance between the centers of the peaks of doublet is called as?
How many peaks are expected in low-resolution NMR spectrum of vinyl chloride and ethyl cyclopropane?
Which of the following phase changes are endothermic?
The Gibbs free energy is positive when a change in enthalpy and change in entropy are positive at ______.
The type of hardness that occurs due to the presence of bicarbonate of calcium and magnesium ions hardness is ______.
Which of the following is an alkane which can exhibit optical activity?
Q.2 Solve both questions :
Write short notes on:
What are Quantum numbers and what do they signify? How are these numbers related with each other?
Q.3 Solve both questions :
Give reason for the following:-
Find out the bond orders of CO, $ \text{N}_2 $, $ \text{O}_2^\text{2-} $, $ \text{O}_2^\text{2+} $
Which of the following two molecules has a higher bond length (i) $ \text{O}_2 $: (ii) $ \text{O}_2^- $? Explain using Molecular orbital Theory.
Q.4 Solve both questions :
Describe and compare the splitting of d- orbitals under the influence of octahedral and tetrahedral ligand fields.
Explain Crystal Field Stabilization Energy (CFSE) and pairing Energy (PE). How do the CFSE values related to the corresponding colours of d-block metal complexes?
Q.5 Solve all parts :
Why is methanol a good solvent for U.V. Visible but not for IR spectroscopy?
Calculate the number of vibrational degrees of freedom in the following compounds
What is the importance of fingerprint region in IR spectra?
Q.6 Solve both questions :
Discuss spin-spin relaxation in NMR spectroscopy and draw and explain the splitting pattern observed in the 1H NMR of $ \text{CHCl}_2\text{CH}_2\text{Cl} $.
The frequency of oscillation of HF is $ 4.12 \times 10^{-14} $ Hz. Calculate the vibrational energy and its force constant. $ M_{H} = 1.0078u $, $ M_{F} = 18.9984u $.
Q.7 Solve both questions :
What are ion exchange Resins? Discuss their application in water softening. How are spent resins regenerated?
9.67ml of EDTA solution consumes exactly 10.2 mg of $ \text{CaCO}_3 $ equivalents. 100 ml of a given Hard water sample consumes 17.5 ml of above EDTA solution. 250 ml of the above hard water sample was boiled completely and boiled off sample consumed 35 ml of above EDTA solution. Calculate the temporary and permanent hardness of the above hardware sample in $ ^{\circ}C $, $ ^{\circ}F $ and as $ \text{CaCO}_3 $ equivalents.
Q.8 Solve all parts :
Differentiate between terms Configuration and Conformation with suitable examples.
Discuss stereochemistry of tartaric acid.
Explain racemisation of optical isomers with suitable examples.
Q.9 Solve both questions :
What do you mean by cyclization and ring opening reactions give two examples of each type of these reactions.
What are carbonium ions? Show hybridisation in the structure of carbonium ion and discuss the stability of primary, secondary and tertiary carbonium ions.
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. I is compulsory.
Q.1 Answer in brief any seven of the following:
State de Broglie's principle.
What is an orbital?
Arrange the covalent bonds C—F, C—Br, C—Cl and C—I in increasing order of strength.
Which of $ \text{NH}_3 $, $ \text{PH}_3 $, $ \text{AsH}_3 $ and $ \text{SbH}_3 $ possesses largest bond angle? Give reason.
Out of $ \text{Cr}^{2+} $ and $ \text{Cr}^{3+} $, which one is stable in aqueous solution?
$ \text{Cu}^+ $ is colourless, but $ \text{Cu}^{2+} $ is coloured. Why?
What is selection rule?
A gas expands against vacuum. What is the work done on it?
What is the condition for a reaction to be in equilibrium?
The presence of $ \text{CO}_2 $ in boiler feed water should be avoided. Why?
Q.2 Solve both questions :
Write short notes on the following:
Discuss photoelectric effect (with mathematical equations) for explaining the particle nature of light.
How many photons of light having a wavelength of 4000 Å are necessary to provide 1 J of energy? $ (h = 6.626 \times 10^{-34} \, \text{J s}) $
Q.3 Solve this question :
Which of the following two molecules has a higher bond length? (i) $ \text{O}_2 $ (ii) $ \text{O}_2^+ $ (iii) $ \text{O}_2^- $ Explain using molecular orbital theory.
Draw the MO energy level diagram for NO molecule. Using this diagram, calculate and explain bond order and magnetic behaviour of (i) NO, (ii) $ \text{NO}^+ $ and (iii) $ \text{NO}^- $.
Q.4 Solve all parts :
The internuclear distance of NaCl is $ 2.36 \times 10^{-10} \text{ m} $. Calculate the reduced mass and moment of inertia of NaCl. (Atomic mass of Cl = $ 35 \times 10^{-3} \text{ kg mol}^{-1} $ and Na = $ 23 \times 10^{-3} \text{ kg mol}^{-1} $).
Calculate the force constant for CO, if it absorbs at $ 2.143 \times 10^{5} \, \text{m}^{-1} $. (Atomic mass of C = $ 12 \times 10^{-3} \, \text{kg mol}^{-1} $ and O = $ 16 \times 10^{-3} \, \text{kg mol}^{-1} $)
Which type of electronic transition(s) is/are observed in UV-visible spectrum of phenol in the range 200 nm to 900 nm? Justify your answer.
Q.5 Solve all parts :
How many types of $ ^{1}\text{H} $ NMR signals are expected for (i) $ \text{CH}_2\text{BrCH}_2\text{Cl} $ and (ii) $ \text{CH}_3\text{OCH}_3 $? Mention relative intensity ratio for the signal(s) observed for (i) and (ii).
At what frequency shift from TMS, would a group of nuclei with δ = 1:5 resonate in NMR spectrometers operating at 60 MHz and 400 MHz?
2 mol of $ \text{NH}_3 $ at 300 K occupy a volume of $ 5 \times 10^{-3} \, \text{m}^3 $. Calculate the pressure using van der Waals equation $ (a = 0.417 \, \text{Nm}^4 \, \text{mol}^{-2} $ and $ b = 0.037 \times 10^{-3} \, \text{m}^3 \, \text{mol}^{-1}) $. Compare the above result with the pressure calculated using ideal gas equation.
Q.6 Solve both questions :
7 mol of a monatomic ideal gas are compressed reversibly and adiabatically. The initial volume is 16 dm³ and the final volume is 7 dm³. The initial temperature is 27 °C.
Given, $ C_v = 20 \cdot 91 \, \text{JK}^{-1} \, \text{mol}^{-1} $, $ \gamma = 1 \cdot 4 $.
Write a cell (in proper cell representation) whose cell reaction is $ \text{AgCl} \rightarrow \text{Ag}^+ + \text{Cl}^- $, using the following standard electrode potentials at 298 K:
$ E°_{\text{AgCl}/\text{AgCl}^-} = 0 \cdot 22 \, \text{V} $, $ E°_{\text{Ag}^+/\text{Ag}} = 0 \cdot 80 \, \text{V} $
Calculate $ E° $ of the cell. Calculate solubility product (or solubility constant) of AgCl and its solubility at 298 K.
Q.7 Solve both questions :
A water sample had the following constituents per litre:
$ \text{CaCO}_3 = 65 \, \text{mg} $, $ \text{MgHCO}_3 = 80 \, \text{mg} $
$ \text{CaSO}_4 = 155 \, \text{mg} $, $ \text{MgSO}_4 = 135 \, \text{mg} $
$ \text{NaCl} = 8 \, \text{mg} $
Calculate the quantity of temporary and permanent hardness in the water sample. Calculate the quantity of lime (80% purity) and soda (90% purity) required for softening of 1 million litre of above water sample.
In an experiment to determine the hardness of a sample of water, 25 ml of N/50 $ \text{Na}_2\text{CO}_3 $ solution was added to 100 ml of water sample. After complete precipitation of insoluble carbonate, the unreacted $ \text{Na}_2\text{CO}_3 $ was titrated against N/50 $ \text{H}_2\text{SO}_4 $ solution, when 10 ml of acid was required. Calculate the hardness and comment on the nature of hardness so determined.
Q.8 Solve both questions :
Write notes on the following:
Differentiate between the following:
Q.9 Solve both questions :
Explain the following:
Arrange the following carbocations in order of increasing stability with suitable reasons:
$ \text{C}_6\text{H}_5\text{CH}_2^+ $
$ (\text{CH}_3)_3\text{C}^+ $
$ (\text{C}_6\text{H}_5)_2\text{CH}^+ $
$ \text{CH}_3\text{CH}_2^+ $
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. I is compulsory.
Q.1 Answer in brief any seven of the following:
State de Broglie's principle.
What is an orbital?
Arrange the covalent bonds C—F, C—Br, C—Cl and C—I in increasing order of strength.
Which of $ \text{NH}_3 $, $ \text{PH}_3 $, $ \text{AsH}_3 $ and $ \text{SbH}_3 $ possesses largest bond angle? Give reason.
Out of $ \text{Cr}^{2+} $ and $ \text{Cr}^{3+} $, which one is stable in aqueous solution?
$ \text{Cu}^+ $ is colourless, but $ \text{Cu}^{2+} $ is coloured. Why?
What is selection rule?
A gas expands against vacuum. What is the work done on it?
What is the condition for a reaction to be in equilibrium?
The presence of $ \text{CO}_2 $ in boiler feed water should be avoided. Why?
Q.2 Solve both questions :
Write short notes on the following:
Discuss photoelectric effect (with mathematical equations) for explaining the particle nature of light.
How many photons of light having a wavelength of 4000 Å are necessary to provide 1 J of energy? $ (h = 6.626 \times 10^{-34} \, \text{J s}) $
Q.3 Solve this question :
Which of the following two molecules has a higher bond length? (i) $ \text{O}_2 $ (ii) $ \text{O}_2^+ $ (iii) $ \text{O}_2^- $ Explain using molecular orbital theory.
Draw the MO energy level diagram for NO molecule. Using this diagram, calculate and explain bond order and magnetic behaviour of (i) NO, (ii) $ \text{NO}^+ $ and (iii) $ \text{NO}^- $.
Q.4 Solve all parts :
The internuclear distance of NaCl is $ 2.36 \times 10^{-10} \text{ m} $. Calculate the reduced mass and moment of inertia of NaCl. (Atomic mass of Cl = $ 35 \times 10^{-3} \text{ kg mol}^{-1} $ and Na = $ 23 \times 10^{-3} \text{ kg mol}^{-1} $).
Calculate the force constant for CO, if it absorbs at $ 2.143 \times 10^{5} \, \text{m}^{-1} $. (Atomic mass of C = $ 12 \times 10^{-3} \, \text{kg mol}^{-1} $ and O = $ 16 \times 10^{-3} \, \text{kg mol}^{-1} $)
Which type of electronic transition(s) is/are observed in UV-visible spectrum of phenol in the range 200 nm to 900 nm? Justify your answer.
Q.5 Solve all parts :
How many types of $ ^{1}\text{H} $ NMR signals are expected for (i) $ \text{CH}_2\text{BrCH}_2\text{Cl} $ and (ii) $ \text{CH}_3\text{OCH}_3 $? Mention relative intensity ratio for the signal(s) observed for (i) and (ii).
At what frequency shift from TMS, would a group of nuclei with δ = 1:5 resonate in NMR spectrometers operating at 60 MHz and 400 MHz?
2 mol of $ \text{NH}_3 $ at 300 K occupy a volume of $ 5 \times 10^{-3} \, \text{m}^3 $. Calculate the pressure using van der Waals equation $ (a = 0.417 \, \text{Nm}^4 \, \text{mol}^{-2} $ and $ b = 0.037 \times 10^{-3} \, \text{m}^3 \, \text{mol}^{-1}) $. Compare the above result with the pressure calculated using ideal gas equation.
Q.6 Solve both questions :
7 mol of a monatomic ideal gas are compressed reversibly and adiabatically. The initial volume is 16 dm³ and the final volume is 7 dm³. The initial temperature is 27 °C.
Given, $ C_v = 20 \cdot 91 \, \text{JK}^{-1} \, \text{mol}^{-1} $, $ \gamma = 1 \cdot 4 $.
Write a cell (in proper cell representation) whose cell reaction is $ \text{AgCl} \rightarrow \text{Ag}^+ + \text{Cl}^- $, using the following standard electrode potentials at 298 K:
$ E°_{\text{AgCl}/\text{AgCl}^-} = 0 \cdot 22 \, \text{V} $, $ E°_{\text{Ag}^+/\text{Ag}} = 0 \cdot 80 \, \text{V} $
Calculate $ E° $ of the cell. Calculate solubility product (or solubility constant) of AgCl and its solubility at 298 K.
Q.7 Solve both questions :
A water sample had the following constituents per litre:
$ \text{CaCO}_3 = 65 \, \text{mg} $, $ \text{MgHCO}_3 = 80 \, \text{mg} $
$ \text{CaSO}_4 = 155 \, \text{mg} $, $ \text{MgSO}_4 = 135 \, \text{mg} $
$ \text{NaCl} = 8 \, \text{mg} $
Calculate the quantity of temporary and permanent hardness in the water sample. Calculate the quantity of lime (80% purity) and soda (90% purity) required for softening of 1 million litre of above water sample.
In an experiment to determine the hardness of a sample of water, 25 ml of N/50 $ \text{Na}_2\text{CO}_3 $ solution was added to 100 ml of water sample. After complete precipitation of insoluble carbonate, the unreacted $ \text{Na}_2\text{CO}_3 $ was titrated against N/50 $ \text{H}_2\text{SO}_4 $ solution, when 10 ml of acid was required. Calculate the hardness and comment on the nature of hardness so determined.
Q.8 Solve both questions :
Write notes on the following:
Differentiate between the following:
Q.9 Solve both questions :
Explain the following:
Arrange the following carbocations in order of increasing stability with suitable reasons:
$ \text{C}_6\text{H}_5\text{CH}_2^+ $
$ (\text{CH}_3)_3\text{C}^+ $
$ (\text{C}_6\text{H}_5)_2\text{CH}^+ $
$ \text{CH}_3\text{CH}_2^+ $
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. 1 is compulsory.
Q.1 Answer any seven questions in brief:
What are the shapes of $ \text{NF}_3 $, $ \text{BF}_3 $ and $ \text{ClF}_3 $ molecules?
How many $ \text{sp}^3 $-, $ \text{sp}^2 $- and sp-hybridized carbon atoms are present in $ (\text{CH}_3)_2\text{CH}-\text{CH}=\text{CH}-\text{C}=\text{C}-\text{CH}=\text{CH}_2 $?
Arrange molecular species $ \text{N}_2 $, $ \text{N}_2^+ $, $ \text{N}_2^- $ and $ \text{N}_2^{2-} $ in increasing order of stability.
Transition metal ions like $ \text{Cu}^+ $ and $ \text{Ag}^+ $ are colourless. Why?
What type of magnetism is exhibited by $ [\text{Mn(H}_2\text{O)}_6]^{2+} $ ion?
How are exhausted zeolite regenerated?
Name any two coagulants.
A gas expands against vacuum. What is the work done on it?
Under what condition, $ \Delta H = \Delta U $?
What type of transition can take place in $ \text{CH}_3\text{NO}_2 $ in UV spectroscopy?
Q.2 Solve all parts :
Discuss photoelectric effect and explain equations involved with this effect.
How many photons of light having a wavelength of 4000 Å are necessary to provide 1 J of energy? $ (h = 6 \cdot 626 \times 10^{-34} \, \text{J-s}) $.
If the electron in an unexcited hydrogen atom is excited by absorption of $ 1.76 \times 10^{-18} \, \text{J} $ per atom, what would be the wavelength of radiation emitted when the atom returns to its ground state? $ (c = 3 \times 10^8 \, \text{m s}^{-1}, \, h = 6 \cdot 626 \times 10^{-34} \, \text{J-s}) $
Q.3 Solve both questions :
Draw the MO energy level diagram for $ \text{B}_2 $ and based on the diagram, explain the magnetic property observed in $ \text{B}_2 $.
Which type of isomerism is observed with the metal complex having molecular formula, $ \text{CoBr(SO}_4) \cdot \text{5NH}_3 $? Draw their structures. Write one chemical test (reaction) in favour of each structure.
Q.4 Solve all parts :
Calculate the frequency (in Hz and cm⁻¹) of O-H bond, if the force constant and reduced mass of the atom pair are 770 N m⁻¹ and $ 1.563 \times 10^{-27} \, \text{kg} $ respectively.
Microwave spectrum of gaseous HCl molecule exhibits a series of equally spaced lines with interspacing of 20-7 cm⁻¹. Calculate the internuclear distance of HCl molecule.
Draw a relative energy diagram showing $ \sigma $, $ \pi $, $ n $, $ \pi^* $ and $ \sigma^* $ energy levels. Using this diagram, discuss the variation of energy levels for $ \pi \rightarrow \pi^* $ and $ n \rightarrow \pi^* $ transitions with increase in solvent polarity qualitatively.
Q.5 Solve all parts :
At what frequency shift from TMS, would a group of nuclei with δ = 7.50 resonate in an NMR spectrometer operating at 500 MHz?
Write the number of $ ^{1}\text{H} $-NMR signals and splitting pattern of the signals (singlet, doublet, etc.) due to spin-spin coupling for the following molecules with proper justification:
Use the equation of state of van der Waals to calculate the pressure of 5 moles of $ \text{NH}_3(g) $ occupying a volume of 7 L at 373 K. (Given, $ a = 4 \cdot 17 \, \text{L}^2 \, \text{atm mol}^{-2} $, $ b = 0 \cdot 0371 \, \text{L mol}^{-1} $). Compare the above result with the pressure calculated using ideal gas equation.
Q.6 Solve both questions :
1 mole of an ideal gas expands isothermally and reversibly from 5 dm³ to 10 dm³ at 300 K. Calculate $ q $, $ w $, $ \Delta U $, $ \Delta H $, $ \Delta G $ and $ \Delta A $.
Design electrochemical cells (in proper cell representation/notation) in which each of the following reactions occurs:
Q.7 Solve all parts :
Write the principle for lime soda process for softening of hard water.
Calculate the amount of lime and soda required for the softening of a million litres of hard water containing $ \text{CaCO}_3 = 25 \, ppm $, $ \text{MgCO}_3 = 144 \, ppm $, $ \text{CaCl}_2 = 111 \, ppm $, $ \text{MgCl}_2 = 95 \, ppm $, $ \text{Na}_2\text{SO}_4 = 15 \, ppm $, $ \text{Fe}_2\text{O}_3 = 25 \, ppm $.
The hardness of 50000 litres of water sample was removed by passing it through a zeolite softener. The softener then required 200 L of NaCl solution, containing 125 g/L of NaCl for regeneration. Calculate the hardness of the sample of water.
Q.8 Solve both questions :
Distinguish between the Pauling and Mulliken electronegativity scales.
Draw all the optical isomers of tartaric acid. Identify the pair(s) of enantiomers and diastereomers from the above optical isomers.
Q.9 Solve both questions :
Explain the following:
0.5 g of $ \text{CaCO}_3 $ was dissolved in HCl and the solution was made up to 500 mL with distilled water. 50 mL of this solution required 48 mL of EDTA solution for titration. 50 mL of water sample required 15 mL of EDTA and after boiling and filtering required 10 mL of EDTA solution. Calculate total hardness, non-carbonate hardness and carbonate hardness of the water sample.
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. 1 is compulsory.
Q.1 Answer any seven questions in brief:
What are the shapes of $ \text{NF}_3 $, $ \text{BF}_3 $ and $ \text{ClF}_3 $ molecules?
How many $ \text{sp}^3 $-, $ \text{sp}^2 $- and sp-hybridized carbon atoms are present in $ (\text{CH}_3)_2\text{CH}-\text{CH}=\text{CH}-\text{C}=\text{C}-\text{CH}=\text{CH}_2 $?
Arrange molecular species $ \text{N}_2 $, $ \text{N}_2^+ $, $ \text{N}_2^- $ and $ \text{N}_2^{2-} $ in increasing order of stability.
Transition metal ions like $ \text{Cu}^+ $ and $ \text{Ag}^+ $ are colourless. Why?
What type of magnetism is exhibited by $ [\text{Mn(H}_2\text{O)}_6]^{2+} $ ion?
How are exhausted zeolite regenerated?
Name any two coagulants.
A gas expands against vacuum. What is the work done on it?
Under what condition, $ \Delta H = \Delta U $?
What type of transition can take place in $ \text{CH}_3\text{NO}_2 $ in UV spectroscopy?
Q.2 Solve all parts :
Discuss photoelectric effect and explain equations involved with this effect.
How many photons of light having a wavelength of 4000 Å are necessary to provide 1 J of energy? $ (h = 6 \cdot 626 \times 10^{-34} \, \text{J-s}) $.
If the electron in an unexcited hydrogen atom is excited by absorption of $ 1.76 \times 10^{-18} \, \text{J} $ per atom, what would be the wavelength of radiation emitted when the atom returns to its ground state? $ (c = 3 \times 10^8 \, \text{m s}^{-1}, \, h = 6 \cdot 626 \times 10^{-34} \, \text{J-s}) $
Q.3 Solve both questions :
Draw the MO energy level diagram for $ \text{B}_2 $ and based on the diagram, explain the magnetic property observed in $ \text{B}_2 $.
Which type of isomerism is observed with the metal complex having molecular formula, $ \text{CoBr(SO}_4) \cdot \text{5NH}_3 $? Draw their structures. Write one chemical test (reaction) in favour of each structure.
Q.4 Solve all parts :
Calculate the frequency (in Hz and cm⁻¹) of O-H bond, if the force constant and reduced mass of the atom pair are 770 N m⁻¹ and $ 1.563 \times 10^{-27} \, \text{kg} $ respectively.
Microwave spectrum of gaseous HCl molecule exhibits a series of equally spaced lines with interspacing of 20-7 cm⁻¹. Calculate the internuclear distance of HCl molecule.
Draw a relative energy diagram showing $ \sigma $, $ \pi $, $ n $, $ \pi^* $ and $ \sigma^* $ energy levels. Using this diagram, discuss the variation of energy levels for $ \pi \rightarrow \pi^* $ and $ n \rightarrow \pi^* $ transitions with increase in solvent polarity qualitatively.
Q.5 Solve all parts :
At what frequency shift from TMS, would a group of nuclei with δ = 7.50 resonate in an NMR spectrometer operating at 500 MHz?
Write the number of $ ^{1}\text{H} $-NMR signals and splitting pattern of the signals (singlet, doublet, etc.) due to spin-spin coupling for the following molecules with proper justification:
Use the equation of state of van der Waals to calculate the pressure of 5 moles of $ \text{NH}_3(g) $ occupying a volume of 7 L at 373 K. (Given, $ a = 4 \cdot 17 \, \text{L}^2 \, \text{atm mol}^{-2} $, $ b = 0 \cdot 0371 \, \text{L mol}^{-1} $). Compare the above result with the pressure calculated using ideal gas equation.
Q.6 Solve both questions :
1 mole of an ideal gas expands isothermally and reversibly from 5 dm³ to 10 dm³ at 300 K. Calculate $ q $, $ w $, $ \Delta U $, $ \Delta H $, $ \Delta G $ and $ \Delta A $.
Design electrochemical cells (in proper cell representation/notation) in which each of the following reactions occurs:
Q.7 Solve all parts :
Write the principle for lime soda process for softening of hard water.
Calculate the amount of lime and soda required for the softening of a million litres of hard water containing $ \text{CaCO}_3 = 25 \, ppm $, $ \text{MgCO}_3 = 144 \, ppm $, $ \text{CaCl}_2 = 111 \, ppm $, $ \text{MgCl}_2 = 95 \, ppm $, $ \text{Na}_2\text{SO}_4 = 15 \, ppm $, $ \text{Fe}_2\text{O}_3 = 25 \, ppm $.
The hardness of 50000 litres of water sample was removed by passing it through a zeolite softener. The softener then required 200 L of NaCl solution, containing 125 g/L of NaCl for regeneration. Calculate the hardness of the sample of water.
Q.8 Solve both questions :
Distinguish between the Pauling and Mulliken electronegativity scales.
Draw all the optical isomers of tartaric acid. Identify the pair(s) of enantiomers and diastereomers from the above optical isomers.
Q.9 Solve both questions :
Explain the following:
0.5 g of $ \text{CaCO}_3 $ was dissolved in HCl and the solution was made up to 500 mL with distilled water. 50 mL of this solution required 48 mL of EDTA solution for titration. 50 mL of water sample required 15 mL of EDTA and after boiling and filtering required 10 mL of EDTA solution. Calculate total hardness, non-carbonate hardness and carbonate hardness of the water sample.
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. 1 is compulsory.
Q.1 Answer any seven questions in brief:
State de Broglie's principle.
What is an orbital?
What are the shapes of $ \text{BF}_3 $ and $ \text{ClF}_3 $ molecules?
Out of $ \text{NH}_3 $, $ \text{PH}_3 $, $ \text{AsH}_2 $ and $ \text{SbH}_2 $, which possesses largest bond angle? Give reason.
Out of $ \text{Cr}^{2+} $ and $ \text{Cr}^{3+} $, which one is stable in aqueous solution?
$ \text{Cu}^+ $ is colourless but $ \text{Cu}^{2+} $ is coloured. Why?
What is selection rule?
A gas expands against vacuum. What is the work done on it?
What is the condition for a reaction to be in equilibrium?
The presence of $ \text{CO}_2 $ in boiler feed water should be avoided. Why?
Q.2 Solve both questions :
A water sample had the following constituents per litre:
$ \text{CaCO}_3 = 160 \, \text{mg} $
$ \text{MgHCO}_3 = 150 \, \text{mg} $
$ \text{CaSO}_4 = 136 \, \text{mg} $
$ \text{MgSO}_4 = 120 \, \text{mg} $
$ \text{NaCl} = 10 \, \text{mg} $
Calculate the quantity of temporary and permanent hardness in the water sample. Calculate the quantity of lime (78% purity) and soda (92% purity) required for softening of 25 million litre of above water sample.
In an experiment to determine the hardness of a sample of water, 50 mL of $ N/50 \, \text{Na}_2\text{CO}_3 $ solution was added to 200 mL of water sample. After complete precipitation of insoluble carbonate, the unreacted $ \text{Na}_2\text{CO}_3 $ was titrated against $ N/50 \, \text{H}_2\text{SO}_4 $ solution, when 20 mL of acid was required. Calculate the hardness and comment on the nature of hardness so determined.
Q.3 Solve both questions :
Write short notes on the following:
How many photons of light having a wavelength of 3000 Å are necessary to provide 1 J of energy? $ (h = 6 \cdot 626 \times 10^{-34} \, \text{J-s}) $
Q.4 Solve both questions :
Explain the behaviour of CO as ligand with different metal ions using molecular orbital theory.
Draw the MO energy-level diagram for NO molecule. Using this diagram, calculate and explain bond order and magnetic behaviour of (i) NO, (ii) $ \text{NO}^+ $ and (iii) $ \text{NO}^- $.
Q.5 Solve all parts :
Which type of electronic transition(s) is/are observed in UV-visible spectrum of aniline in the range 200 nm to 900 nm? Justify your answer with suitable figure.
The internuclear distance of NaCl is $ 2 \cdot 36 \times 10^{-10} \, \text{m} $. Calculate the reduced mass and moment of inertia of NaCl. (Atomic mass of Cl = $ 35 \times 10^{-3} \, \text{kg mol}^{-1} $ and Na = $ 23 \times 10^{-3} \, \text{kg mol}^{-1} $)
Calculate the force constant for CO, if it absorbs at $ 2 \cdot 143 \times 10^{5} \, \text{m}^{-1} $. (Atomic mass of C = $ 12 \times 10^{-3} \, \text{kg mol}^{-1} $ and O = $ 16 \times 10^{-3} \, \text{kg mol}^{-1} $)
Q.6 Solve both questions :
How many types of $ ^{1}\text{H} $ NMR signals are expected for (i) $ \text{CH}_3\text{COOCH}_2\text{CH}_3 $ and (ii) $ \text{CH}_3\text{CHCl}_2 $? Mention the relative intensity ratio for the signal(s) observed for (i) and (ii).
0-6 mol of $ \text{NH}_3 $ at 25°C occupies a volume of 3 dm³. Calculate the pressure using van der Waals equation $ (a = 0.417 \, \text{N m}^4 \, \text{mol}^{-2} $ and $ b = 0.037 \times 10^{-3} \, \text{m}^3 \, \text{mol}^{-1}) $. Compare the above result with the pressure calculated using ideal gas equation.
Q.7 Solve both questions :
7 mol of a monatomic ideal gas are compressed reversibly and adiabatically. The initial volume is 15 dm³ and the final volume is 9 dm³. The initial temperature is 27 °C. (i) What would be the final temperature in this process? (ii) Calculate w, q and ΔU for the process. Given, $ C_v = 20 \cdot 91 \, \text{J K}^{-1} \, \text{mol}^{-1} $ and $ \gamma = 1 \cdot 4 $.
Write a cell representation (in proper cell representation) whose cell reaction is $ \text{AgCl} \rightarrow \text{Ag}^+ + \text{Cl}^- $, using the following standard electrode potentials at 298 K:
$ E_{\text{AgCl}/\text{Ag, Cl}}^o = 0 \cdot 22 \, \text{V} $
$ E_{\text{Ag}^+/\text{Ag}}^o = 0 \cdot 80 \, \text{V} $
Calculate $ E^o $ of the cell. Calculate solubility product (or solubility constant) of AgCl and its solubility at 298 K.
Q.8 Solve both questions :
Write notes on the following:
Differentiate between the following:
Q.9 Solve both questions :
Explain the following:
Arrange the following carbocations in order of increasing stability with suitable reasons:
$ \text{C}_6\text{H}_5\text{CH}_2^+ $
$ (\text{CH}_3)_3\text{C}^+ $
$
(\text{C}_6\text{H}_5)_2\text{CH}^+ $
$ \text{CH}_3\text{CH}_2^+ $
Write short notes on the following:
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. 1 is compulsory.
Q.1 Answer any seven questions in brief:
State de Broglie's principle.
What is an orbital?
What are the shapes of $ \text{BF}_3 $ and $ \text{ClF}_3 $ molecules?
Out of $ \text{NH}_3 $, $ \text{PH}_3 $, $ \text{AsH}_2 $ and $ \text{SbH}_2 $, which possesses largest bond angle? Give reason.
Out of $ \text{Cr}^{2+} $ and $ \text{Cr}^{3+} $, which one is stable in aqueous solution?
$ \text{Cu}^+ $ is colourless but $ \text{Cu}^{2+} $ is coloured. Why?
What is selection rule?
A gas expands against vacuum. What is the work done on it?
What is the condition for a reaction to be in equilibrium?
The presence of $ \text{CO}_2 $ in boiler feed water should be avoided. Why?
Q.2 Solve both questions :
A water sample had the following constituents per litre:
$ \text{CaCO}_3 = 160 \, \text{mg} $
$ \text{MgHCO}_3 = 150 \, \text{mg} $
$ \text{CaSO}_4 = 136 \, \text{mg} $
$ \text{MgSO}_4 = 120 \, \text{mg} $
$ \text{NaCl} = 10 \, \text{mg} $
Calculate the quantity of temporary and permanent hardness in the water sample. Calculate the quantity of lime (78% purity) and soda (92% purity) required for softening of 25 million litre of above water sample.
In an experiment to determine the hardness of a sample of water, 50 mL of $ N/50 \, \text{Na}_2\text{CO}_3 $ solution was added to 200 mL of water sample. After complete precipitation of insoluble carbonate, the unreacted $ \text{Na}_2\text{CO}_3 $ was titrated against $ N/50 \, \text{H}_2\text{SO}_4 $ solution, when 20 mL of acid was required. Calculate the hardness and comment on the nature of hardness so determined.
Q.3 Solve both questions :
Write short notes on the following:
How many photons of light having a wavelength of 3000 Å are necessary to provide 1 J of energy? $ (h = 6 \cdot 626 \times 10^{-34} \, \text{J-s}) $
Q.4 Solve both questions :
Explain the behaviour of CO as ligand with different metal ions using molecular orbital theory.
Draw the MO energy-level diagram for NO molecule. Using this diagram, calculate and explain bond order and magnetic behaviour of (i) NO, (ii) $ \text{NO}^+ $ and (iii) $ \text{NO}^- $.
Q.5 Solve all parts :
Which type of electronic transition(s) is/are observed in UV-visible spectrum of aniline in the range 200 nm to 900 nm? Justify your answer with suitable figure.
The internuclear distance of NaCl is $ 2 \cdot 36 \times 10^{-10} \, \text{m} $. Calculate the reduced mass and moment of inertia of NaCl. (Atomic mass of Cl = $ 35 \times 10^{-3} \, \text{kg mol}^{-1} $ and Na = $ 23 \times 10^{-3} \, \text{kg mol}^{-1} $)
Calculate the force constant for CO, if it absorbs at $ 2 \cdot 143 \times 10^{5} \, \text{m}^{-1} $. (Atomic mass of C = $ 12 \times 10^{-3} \, \text{kg mol}^{-1} $ and O = $ 16 \times 10^{-3} \, \text{kg mol}^{-1} $)
Q.6 Solve both questions :
How many types of $ ^{1}\text{H} $ NMR signals are expected for (i) $ \text{CH}_3\text{COOCH}_2\text{CH}_3 $ and (ii) $ \text{CH}_3\text{CHCl}_2 $? Mention the relative intensity ratio for the signal(s) observed for (i) and (ii).
0-6 mol of $ \text{NH}_3 $ at 25°C occupies a volume of 3 dm³. Calculate the pressure using van der Waals equation $ (a = 0.417 \, \text{N m}^4 \, \text{mol}^{-2} $ and $ b = 0.037 \times 10^{-3} \, \text{m}^3 \, \text{mol}^{-1}) $. Compare the above result with the pressure calculated using ideal gas equation.
Q.7 Solve both questions :
7 mol of a monatomic ideal gas are compressed reversibly and adiabatically. The initial volume is 15 dm³ and the final volume is 9 dm³. The initial temperature is 27 °C. (i) What would be the final temperature in this process? (ii) Calculate w, q and ΔU for the process. Given, $ C_v = 20 \cdot 91 \, \text{J K}^{-1} \, \text{mol}^{-1} $ and $ \gamma = 1 \cdot 4 $.
Write a cell representation (in proper cell representation) whose cell reaction is $ \text{AgCl} \rightarrow \text{Ag}^+ + \text{Cl}^- $, using the following standard electrode potentials at 298 K:
$ E_{\text{AgCl}/\text{Ag, Cl}}^o = 0 \cdot 22 \, \text{V} $
$ E_{\text{Ag}^+/\text{Ag}}^o = 0 \cdot 80 \, \text{V} $
Calculate $ E^o $ of the cell. Calculate solubility product (or solubility constant) of AgCl and its solubility at 298 K.
Q.8 Solve both questions :
Write notes on the following:
Differentiate between the following:
Q.9 Solve both questions :
Explain the following:
Arrange the following carbocations in order of increasing stability with suitable reasons:
$ \text{C}_6\text{H}_5\text{CH}_2^+ $
$ (\text{CH}_3)_3\text{C}^+ $
$
(\text{C}_6\text{H}_5)_2\text{CH}^+ $
$ \text{CH}_3\text{CH}_2^+ $
Write short notes on the following:
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. I is compulsory.
Q.1 Answer any seven questions in brief:
Arrange the following in increasing order of stability:
$ \text{N}_2, \text{N}_2^+, \text{N}_2^-, \text{N}_2^{2-} $
Transition metal ions like $ \text{Cu}^+ $ and $ \text{Ag}^+ $ are colourless. Why?
Which of $ \text{Cr}^+ $ or $ \text{Cu}^+ $ is expected to be coloured?
$ ^{13}\text{C} $ is NMR active, but $ ^{12}\text{C} $ is not. Why?
What is the direction of a reaction when $ \Delta G = 0 $?
Why is work not a state function?
Write the relationship between parts per million [ppm] and Clarke's degree [°C].
What is critical temperature of a gas?
Arrange the following ligands in order of increasing field strength:
$ \text{CN}^-, \text{CO}, \text{H}_2\text{O}, \text{NH}_3 $
Arrange the following in order of their increasing reactivity in nucleophilic substitution reaction:
$ \text{CH}_3\text{F}, \text{CH}_3\text{I}, \text{CH}_3\text{Br}, \text{CH}_3\text{Cl} $
Q.2 Solve all parts :
At what temperature will water boil when the applied pressure is 528 mm of Hg? (Latent heat of vaporisation of water = 545.5 cal/g)
At NTP, 2.8 L of $ \text{O}_2 $ were mixed with 19.6 L of $ \text{H}_2 $. Calculate the increase in entropy (assume ideal gas behaviour).
The equilibrium constants for the reaction $ \text{H}_2[g] + \text{S(s)} \rightarrow \text{H}_2\text{S[g]} $ are 18.5 at 925 K and 9.25 at 1000 K. Calculate standard enthalpy of the reaction. Also calculate $ \Delta G^\circ $ and $ \Delta S^\circ $ at 925 K.
Q.3 Solve all parts :
The uncertainties in the position and velocity of a particle are $ 9.5 \times 10^{-10} \, \text{m} $ and $ 5.5 \times 10^{-20} \, \text{ms}^{-1} $, respectively. Calculate the mass of the particle. $ (h = 6.626 \times 10^{-34} \, \text{J·s}) $
Calculate the kinetic energy of a moving electron which has a wavelength of 48 pm. [Mass of electron = $ 9.11 \times 10^{-31} \, \text{kg} $]
Discuss the failures of classical mechanics to explain properties of particles at atomic and sub-atomic levels.
Q.4 Solve both questions :
Draw the MO energy-level diagram for $ \text{O}_2 $ and based on the diagram, and explain the magnetic property observed in $ \text{O}_2 $, $ \text{O}_2^+ $ and $ \text{O}_2^- $.
Explain geometrical isomerism and optical isomerism for transition metal complex with an example for each.
Q.5 Solve all parts :
The internuclear distance of NaCl is $ 2.36 \times 10^{-10} \, \text{m} $. Calculate the reduced mass and moment of inertia of NaCl. (Atomic mass of Cl = $ 35 \times 10^{-3} \, \text{kg mol}^{-1} $ and Na = $ 23 \times 10^{-3} \, \text{kg mol}^{-1} $)
Calculate the force constant for CO, if it absorbs at $ 2.143 \times 10^{5} \, \text{m}^{-1} $. (Atomic mass of C = $ 12 \times 10^{-3} \, \text{kg mol}^{-1} $ and O = $ 16 \times 10^{-3} \, \text{kg mol}^{-1} $)
How many $ ^{1}\text{H} $ NMR signals are there in—
Q.6 Solve both questions :
2 mole of $ \text{NH}_3 $ at 300 K occupy a volume of $ 5 \times 10^{-3} \, \text{m}^3 $. Calculate the pressure using van der Waals equation $ (a = 0.417 \, \text{N m}^4 \, \text{mol}^{-2} $ and $ b = 0.037 \times 10^{-3} \, \text{m}^3 \, \text{mol}^{-1}) $. Compare the above result with the pressure calculated using ideal gas equation.
Write short notes on the following:
Q.7 Solve both questions :
Consider the following half-cell reactions:
$ \text{PbO}_2(s) + 4 \text{H}^+ (aq) + \text{SO}_4^{2-} (aq) + 2e \rightarrow $
$ \text{PbSO}_4(s) + 2\text{H}_2\text{O}, E^\circ = 1.70 \, \text{V} $
$ \text{PbSO}_4(s) + 2e \rightarrow \text{Pb}(s) + \text{SO}_4^{2-} (aq) $, $ E^\circ = -0.31 \, \text{V} $
Write the cell (in proper cell notation) and the cell reaction. Calculate the value of $ E^\circ $ for the cell and the EMF generated if $ [\text{H}^+] = 0.1 \, \text{M} $ and $ [\text{SO}_4^{2-}] = 2 \, \text{M} $.
A water sample had the following constituents per litre:
$ \text{CaCO}_3 = 81 \, \text{mg} $, $ \text{MgHCO}_3 = 75 \, \text{mg} $, $ \text{CaSO}_4 = 136 \, \text{mg} $, $ \text{MgSO}_4 = 120 \, \text{mg} $, $ \text{NaCl} = 47 \, \text{mg} $
Calculate the quantity of temporary and permanent hardness in the water sample. Calculate the quantity of lime (78% purity) and soda (92% purity) required for softening of 1.5 million litres of the above water sample.
Q.8 Solve all parts :
Describe two methods used for resolving racemic mixtures into optically active compounds.
Write the possible optical isomers of tartaric acid and indicate the point of symmetry or plane of symmetry (if any) in the isomers.
Differentiate between (i) enantiomers and diastereomers and (ii) racemic mixture and meso compounds.
Q.9 Solve all parts :
How do you decide whether the reaction $ \text{CH}_3\text{Br} + \text{OH}^- \rightleftharpoons \text{CH}_3\text{OH} + \text{Br}^- $ proceeds by $ S_N1 $ or $ S_N2 $ reaction? Give justification in favour of your answer.
Draw the energy profile diagram for the following reaction:
$ (\text{CH}_3)_2\text{CBr} + \text{OH}^- \rightleftharpoons (\text{CH}_3)_2\text{COH} + \text{Br}^- $
Write short notes on the following:
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. I is compulsory.
Q.1 Answer any seven questions in brief:
Arrange the following in increasing order of stability:
$ \text{N}_2, \text{N}_2^+, \text{N}_2^-, \text{N}_2^{2-} $
Transition metal ions like $ \text{Cu}^+ $ and $ \text{Ag}^+ $ are colourless. Why?
Which of $ \text{Cr}^+ $ or $ \text{Cu}^+ $ is expected to be coloured?
$ ^{13}\text{C} $ is NMR active, but $ ^{12}\text{C} $ is not. Why?
What is the direction of a reaction when $ \Delta G = 0 $?
Why is work not a state function?
Write the relationship between parts per million [ppm] and Clarke's degree [°C].
What is critical temperature of a gas?
Arrange the following ligands in order of increasing field strength:
$ \text{CN}^-, \text{CO}, \text{H}_2\text{O}, \text{NH}_3 $
Arrange the following in order of their increasing reactivity in nucleophilic substitution reaction:
$ \text{CH}_3\text{F}, \text{CH}_3\text{I}, \text{CH}_3\text{Br}, \text{CH}_3\text{Cl} $
Q.2 Solve all parts :
At what temperature will water boil when the applied pressure is 528 mm of Hg? (Latent heat of vaporisation of water = 545.5 cal/g)
At NTP, 2.8 L of $ \text{O}_2 $ were mixed with 19.6 L of $ \text{H}_2 $. Calculate the increase in entropy (assume ideal gas behaviour).
The equilibrium constants for the reaction $ \text{H}_2[g] + \text{S(s)} \rightarrow \text{H}_2\text{S[g]} $ are 18.5 at 925 K and 9.25 at 1000 K. Calculate standard enthalpy of the reaction. Also calculate $ \Delta G^\circ $ and $ \Delta S^\circ $ at 925 K.
Q.3 Solve all parts :
The uncertainties in the position and velocity of a particle are $ 9.5 \times 10^{-10} \, \text{m} $ and $ 5.5 \times 10^{-20} \, \text{ms}^{-1} $, respectively. Calculate the mass of the particle. $ (h = 6.626 \times 10^{-34} \, \text{J·s}) $
Calculate the kinetic energy of a moving electron which has a wavelength of 48 pm. [Mass of electron = $ 9.11 \times 10^{-31} \, \text{kg} $]
Discuss the failures of classical mechanics to explain properties of particles at atomic and sub-atomic levels.
Q.4 Solve both questions :
Draw the MO energy-level diagram for $ \text{O}_2 $ and based on the diagram, and explain the magnetic property observed in $ \text{O}_2 $, $ \text{O}_2^+ $ and $ \text{O}_2^- $.
Explain geometrical isomerism and optical isomerism for transition metal complex with an example for each.
Q.5 Solve all parts :
The internuclear distance of NaCl is $ 2.36 \times 10^{-10} \, \text{m} $. Calculate the reduced mass and moment of inertia of NaCl. (Atomic mass of Cl = $ 35 \times 10^{-3} \, \text{kg mol}^{-1} $ and Na = $ 23 \times 10^{-3} \, \text{kg mol}^{-1} $)
Calculate the force constant for CO, if it absorbs at $ 2.143 \times 10^{5} \, \text{m}^{-1} $. (Atomic mass of C = $ 12 \times 10^{-3} \, \text{kg mol}^{-1} $ and O = $ 16 \times 10^{-3} \, \text{kg mol}^{-1} $)
How many $ ^{1}\text{H} $ NMR signals are there in—
Q.6 Solve both questions :
2 mole of $ \text{NH}_3 $ at 300 K occupy a volume of $ 5 \times 10^{-3} \, \text{m}^3 $. Calculate the pressure using van der Waals equation $ (a = 0.417 \, \text{N m}^4 \, \text{mol}^{-2} $ and $ b = 0.037 \times 10^{-3} \, \text{m}^3 \, \text{mol}^{-1}) $. Compare the above result with the pressure calculated using ideal gas equation.
Write short notes on the following:
Q.7 Solve both questions :
Consider the following half-cell reactions:
$ \text{PbO}_2(s) + 4 \text{H}^+ (aq) + \text{SO}_4^{2-} (aq) + 2e \rightarrow $
$ \text{PbSO}_4(s) + 2\text{H}_2\text{O}, E^\circ = 1.70 \, \text{V} $
$ \text{PbSO}_4(s) + 2e \rightarrow \text{Pb}(s) + \text{SO}_4^{2-} (aq) $, $ E^\circ = -0.31 \, \text{V} $
Write the cell (in proper cell notation) and the cell reaction. Calculate the value of $ E^\circ $ for the cell and the EMF generated if $ [\text{H}^+] = 0.1 \, \text{M} $ and $ [\text{SO}_4^{2-}] = 2 \, \text{M} $.
A water sample had the following constituents per litre:
$ \text{CaCO}_3 = 81 \, \text{mg} $, $ \text{MgHCO}_3 = 75 \, \text{mg} $, $ \text{CaSO}_4 = 136 \, \text{mg} $, $ \text{MgSO}_4 = 120 \, \text{mg} $, $ \text{NaCl} = 47 \, \text{mg} $
Calculate the quantity of temporary and permanent hardness in the water sample. Calculate the quantity of lime (78% purity) and soda (92% purity) required for softening of 1.5 million litres of the above water sample.
Q.8 Solve all parts :
Describe two methods used for resolving racemic mixtures into optically active compounds.
Write the possible optical isomers of tartaric acid and indicate the point of symmetry or plane of symmetry (if any) in the isomers.
Differentiate between (i) enantiomers and diastereomers and (ii) racemic mixture and meso compounds.
Q.9 Solve all parts :
How do you decide whether the reaction $ \text{CH}_3\text{Br} + \text{OH}^- \rightleftharpoons \text{CH}_3\text{OH} + \text{Br}^- $ proceeds by $ S_N1 $ or $ S_N2 $ reaction? Give justification in favour of your answer.
Draw the energy profile diagram for the following reaction:
$ (\text{CH}_3)_2\text{CBr} + \text{OH}^- \rightleftharpoons (\text{CH}_3)_2\text{COH} + \text{Br}^- $
Write short notes on the following:
Instructions:
- The marks are indicated in the right-hand margin.
- There are NINE questions in this paper.
- Attempt FIVE questions in all.
- Question No. 1 is compulsory.
Section A
Fill in the blanks/Answer any seven of the following :
Hardness of sample water containing and is —— p.p.m.
Terylene is condensation polymer of ——.
What is Pilling-Bedworth law?
Colligative properties of solution depend ——.
Why boiling point of water increases when NaCl is added?
Why small anodic area results in intense corrosion?
Gutta-percha is polymer of ——.
Define octane number.
Aluminium vessels are used to store conc. . Explain.
Answer the following:
Write the principle of lime-soda process of softening of hard water.
What are the causes of boiler corrosion? How is it controlled?
A of water sample is boiled with solution. The resultant solution is cooled and filtered. The filtrate required solution for complete neutralization. Calculate the hardness of water sample.
Answer the following:
What is Raoult's law? Deduce the relation between relative lowering of vapour pressure and osmotic pressure.
A 3.4% solution of silver nitrate is isotonic with sucrose solution. Calculate the degree of dissociation of silver nitrate.
Answer the following:
Differentiate between thermosetting and thermoplastic resins.
Describe the free radical polymerization mechanism.
Write the preparation and uses of the following : (i) Buna-S (ii) ABS polymer (iii) Nylon.
Answer the following:
Explain carbonization of coal.
Compare the water gas and producer gas in terms of production, composition and calorific value.
How is calorific value of a solid determined by bomb calorimeter?
Answer the following:
What is electrode potential and e.m.f of cell?
Define glass transition temperature.
A of water sample required solution for titration ($1 \text{ ml EDTA} \equiv 1.11 \text{ mg } \text{CaCl}_2$). Calculate the hardness of the sample water.
Answer the following:
Discuss the mechanism of dry and wet corrosion.
What are the factors that affect the rate of corrosion?
Describe sacrificial anodic protection method of controlling corrosion.
Describe the methods of prevention of the following :
Scale and sludge formation
Caustic embrittlement
Priming and foaming
Knocking
Write short notes on :
Water line corrosion
Crevices corrosion
Galvanic series
van't Hoff factor